
In the last video, we talked about collision theory, and we said that molecules must collide to react, and we also said those collisions must have the correct orientation in space to be effective collisions, and finally, those collisions must have enough energy for the reaction to occur. And these ideas of collision theory are contained in the Arrhenius equation. So down here is our equation, where k is our rate constant. So k is the rate constant, the one we talk about in our rate laws. A is called the frequency factor. So, A is the frequency factor. Anybizsoft pdf converter full crack internet. Also called the pre-exponential factor, and A includes things like the frequency of our collisions, and also the orientation of those collisions. And then over here on the right, this e to the negative Ea over RT, this is talking about the fraction of collisions with enough energy for a reaction to occur. This activation energy calculator (also called the Arrhenius equation calculator) can help you calculate the minimum energy required for a chemical reaction to happen. For example, you may want to know what is the energy needed to light a match. In 1889, a Swedish scientist named Svante Arrhenius proposed an equation that relates these concepts with the rate constant: where k represents the rate constant, E a is the activation energy, R is the gas constant (8.3145 J/K mol), and T is the temperature expressed in Kelvin. When the lnk (rate constant) is plotted versus the inverse of the temperature (kelvin), the slope is a straight line. The value of the slope (m) is equal to -Ea/R where R is a constant equal to 8.314 J/mol-K. 'Two-Point Form' of the Arrhenius Equation. The activation energy is the difference in energy between the transition state and the starting reagents. Arrhenius equation [ change| change source ] This equation is a way of calculating the activation energy. So we symbolize this by lowercase f. So the fraction of collisions with enough energy for the reaction to occur. F depends on the activation energy, Ea, which needs to be in joules per mole. R is the gas constant, and T is the temperature in Kelvin. So let's see how changing the activation energy or changing the temperature for a reaction, we'll see how that affects the fraction of collisions with enough energy for our reaction to occur. So, let's start with an activation energy of 40 kJ/mol, and the temperature is 373 K. So, let's solve for f. So, f is equal to e to the negative of our activation energy in joules per mole. So we need to convert 40 kilojoules per mole into joules per mole, so that would be 40,000. So, 40,000 joules per mole. All right, this is over our gas constant, R, and R is equal to 8.314 joules over K times moles. All right, and then this is going to be multiplied by the temperature, which is 373 Kelvin. So let's go ahead and do this calculation, and see what we get. So, let's take out the calculator. E, e to the, we have -40,000, one, two, three divided by 8.314 times 373. So, we get 2.5 times 10 to the -6. So this is equal to 2.5 times 10 to the -6. So what does this mean? Arrhenius Equation Pdf
All right, well, let's say we had one millions collisions. All right, so 1,000,000 collisions. What number divided by 1,000,000, is equal to 2.5 x 10 to the -6? 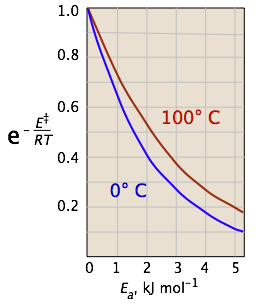
So this number is 2.5. 2. Learning and practicing econometrics solution manual. 5 divided by 1,000,000 is equal to 2.5 x 10 to the -6. So what this means is for every one million collisions in our reaction, only 2.5 collisions have enough energy to react. So obviously that's an extremely small number of collisions with enough energy. All right, let's see what happens when we change the activation energy. So we're going to change the activation energy from 40 kilojoules per mole to 10 kilojoules per mole. So, we're decreasing the activation energy. We're keeping the temperature the same. So let's see how that affects f. So let's plug in this time for f. So f is equal to e to the now we would have -10,000. So we've changed our activation energy, and we're going to divide that by 8.314 times 373. 
So let's do this calculation. So now we have e to the - 10,000 divided by 8.314 times 373. And here we get.04. 
So this is equal to.04. Notice what we've done, we've increased f. We've gone from f equal to 2.5 times 10 to the -6, to.04. So let's stick with this same idea of one million collisions. So let's say, once again, if we had one million collisions here. So 1,000,000 collisions. Arrhenius Equation ProblemsWhat number divided by 1,000,000 is equal to.04? So that number would be 40,000. 40,000 divided by 1,000,000 is equal to.04. So for every one million collisions that we have in our reaction this time 40,000 collisions have enough energy to react, and so that's a huge increase. Right, it's a huge increase in f. It's a huge increase in the number of collisions with enough energy to react, and we did that by decreasing the activation energy. So decreasing the activation energy increased the value for f. Adobe encore preset menus download lagu. It increased the number of effective collisions. All right, let's do one more calculation. This time we're gonna change the temperature. So let's keep the same activation energy as the one we just did. So 10 kilojoules per mole. So 10 kilojoules per mole. This time, let's change the temperature. Here we had 373, let's increase the temperature to 473, and see how that affects the value for f. So f is equal to e to the negative this would be 10,000 again. E to the -10,000 divided by 8.314 times, this time it would 473. So times 473. So let's do this calculation. So e to the -10,000 divided by 8.314 times 473, this time. So we get, let's just say that's.08. So I'll round up to.08 here. So this is equal to.08. So we've increased the value for f, right, we went from.04 to.08, and let's keep our idea of one million collisions. Right, so it's a little bit easier to understand what this means. So what number divided by 1,000,000 is equal to.08. That must be 80,000. Right, so this must be 80,000. So for every 1,000,000 collisions that we have in our reaction, now we have 80,000 collisions with enough energy to react. So we've increased the temperature. Arrhenius Equation Activation Energy Derivation DefinitionGone from 373 to 473. We increased the number of collisions with enough energy to react. We increased the value for f. Finally, let's think about what these things do to the rate constant. So we go back up here to our equation, right, and we've been talking about, well we talked about f.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
